
This correction corresponds to an energy-weighted steepest descent step, and substantially improves the quality of PAO energies. Does it just mean look at the character table (D3h). Ive answered this part of the question, but Im not sure I answered the question the problem was asking. To correct a PAO-HF calculation for residual deficiencies, a noniterative second order correction is introduced. I have a question that asks me to divide the d orbitals of the metal atom in the trigonal prismatic ML6 complex into their symmetry types. For example, Figure: MO Energy Levels (below). The use of augmented functions is recommended. A MO diagram shows the relative energies of MOs and AOs and indicates which MOs form from overlap of which AOs. The results show that the quality of PAO calculations converges smoothly with X. To test the rate of convergence, PAO-HF calculations were performed using cc-pVXZ and aug-cc-pVXZ basis sets for the water monomer and dimer, and six substituted ethylenes. This approximation yields a higher energy than a HF calculation performed in the extended basis, although the two results converge to the same limit as the extended basis approaches completeness on each atom.  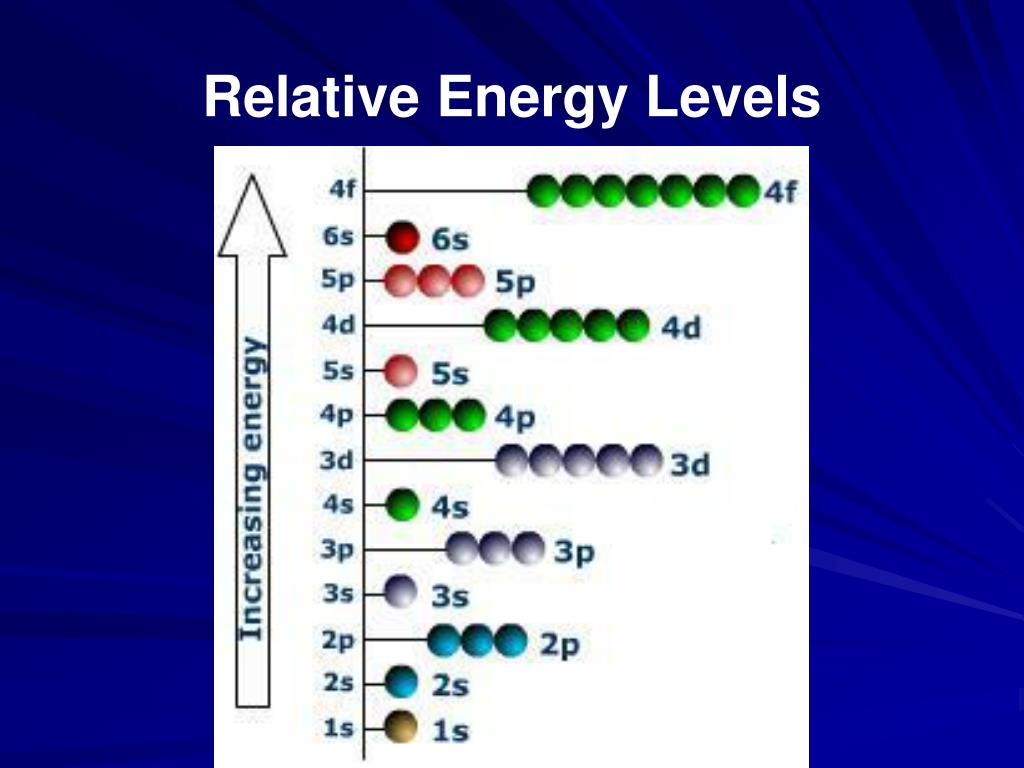
In which cases n+ l values are same, lower n value gets lower energy. Polarized atomic orbitals (PAO’s) are molecule-adapted minimal basis functions that are variationally obtained as an atom-blocked transformation from a conventional extended basis set, as a Hartree–Fock calculation is performed in the PAO basis. The order of relative energies of orbitals is determined by n+l value.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
